The damage from smoking has long been traced through the lungs, heart, and blood vessels. But another pathway may be helping drive harm much farther up the body, into the brain.
A new study from the University of Chicago points to a previously unmapped line of communication between the lungs and the nervous system. This line begins with rare airway cells and ends with changes tied to neurodegeneration. The study, published in Science Advances, suggests nicotine can push specialized lung cells to send out tiny biological packets that disrupt iron balance in nearby neurons. This sets off stress inside the cells and triggers warning signs often linked to diseases such as Alzheimer’s and Parkinson’s.
“This research establishes a clear ‘lung-brain’ axis that helps explain why cigarette smoking is linked to cognitive decline and neurodegenerative risks,” said UChicago postdoctoral researcher Kui Zhang, co-first author of the new work. “By understanding how these exosomes perturb iron homeostasis, we open new doors for protecting neurons from smoke-induced damage.”
That matters because smoking’s connection to dementia has been hard to pin down, even though the link has been reported for years. One study from 2011 found heavy smoking in midlife was tied to more than double the later risk of dementia, Alzheimer’s disease, and vascular dementia. Yet dementia remains less studied as a smoking-related outcome for one grim reason. Many smokers die earlier, before those diseases fully emerge.

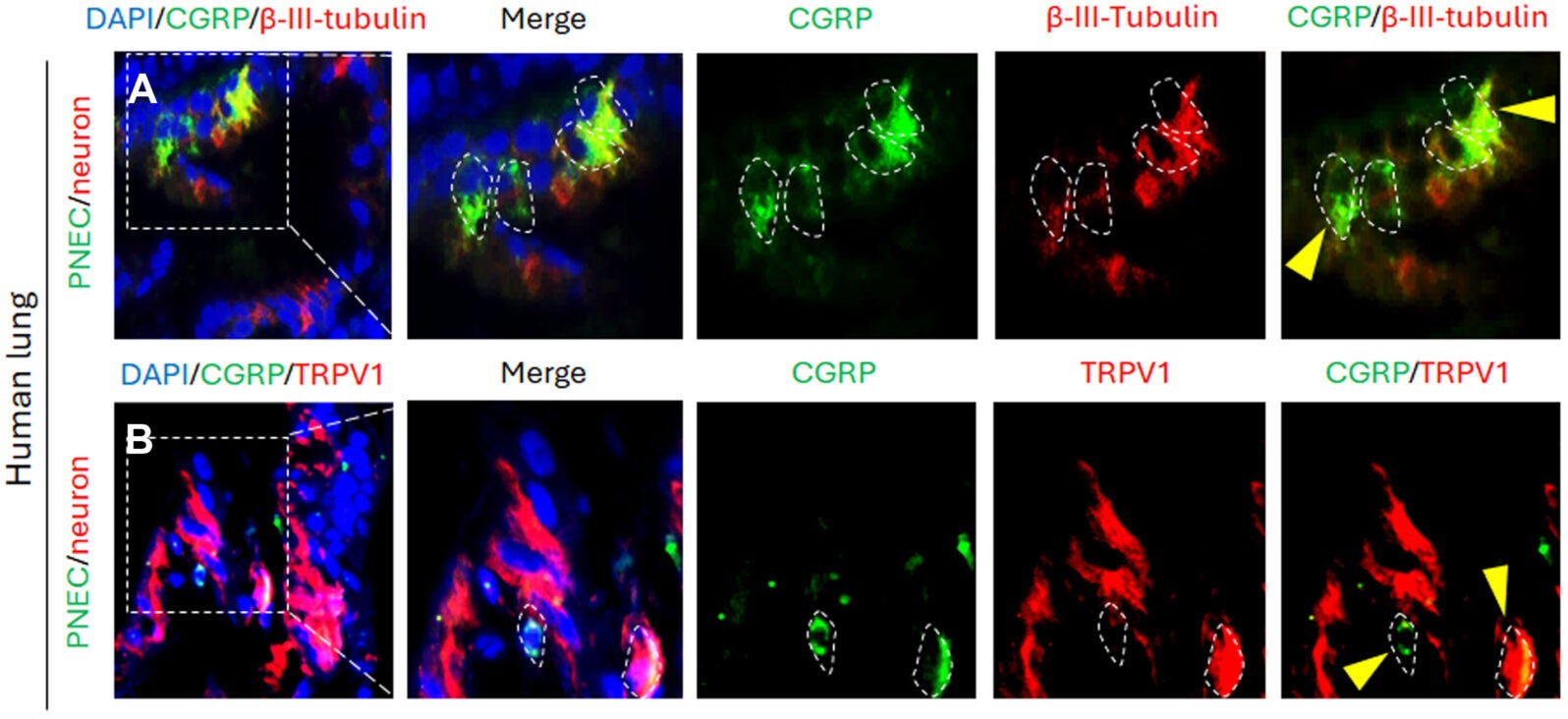
The new research centers on pulmonary neuroendocrine cells, or PNECs, a tiny and unusual cell population in the lungs. These cells make up less than 1% of lung cells. They act as airway sensors, carrying traits of both nerve cells and hormone-releasing cells.
They are also notoriously difficult to study.
“The primary challenge was the extreme rarity of PNECs, which make up less than 1% of lung cells, making them nearly impossible to isolate and study in depth,” Zhang said.
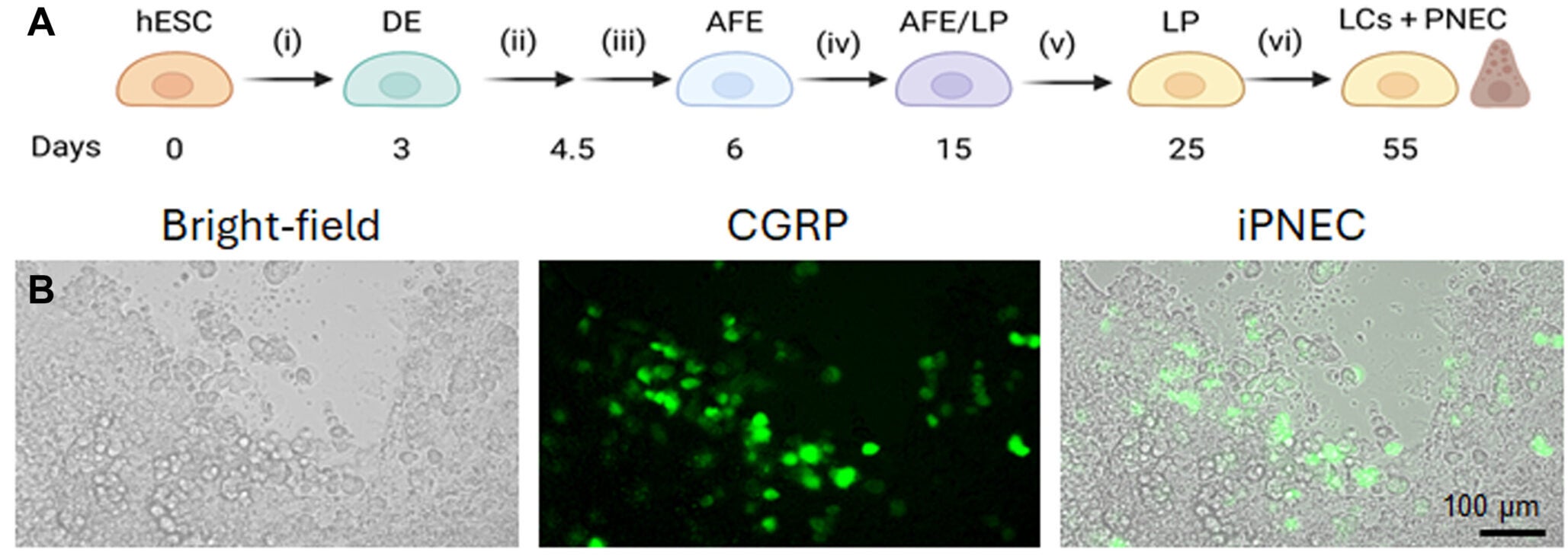
To get around that, the researchers generated induced PNECs from human pluripotent stem cells. That gave them enough cells to test in the lab and compare with native lung tissue.
The team confirmed that PNECs sit close to neurons in both human and mouse lungs. That physical arrangement raised an obvious question. If these cells are so close together, what exactly are they saying to each other?
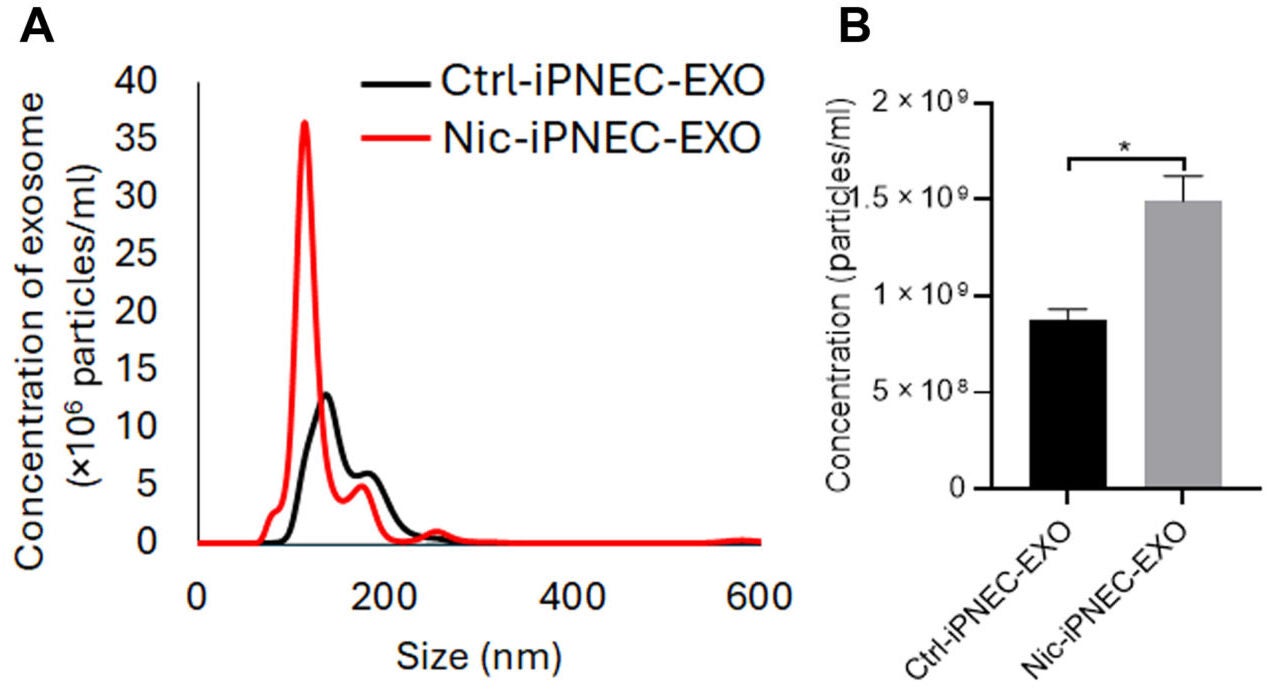
When the induced PNECs were exposed to nicotine, they released far more exosomes, small membrane-bound particles that cells use to shuttle molecular cargo. Exosome release rose 2.8-fold compared with untreated cells. Those nicotine-triggered exosomes were loaded with serotransferrin. This is a protein involved in moving iron through the body.
The finding gave the researchers a possible mechanism. Instead of damage coming only from long-term oxygen loss or vascular strain, nicotine exposure may also spark a biological message from the lung itself.
The problem, the researchers found, is that this message appears to be the wrong one.
When neurons were exposed to exosomes from nicotine-treated PNECs, several proteins involved in iron transport climbed, including TFR1, DMT1, and DCYTB. Ferritin levels rose. ATP, a key energy marker, dropped. Oxidative stress increased. Mitochondrial respiration weakened. The neurons also showed higher levels of alpha-synuclein, a protein strongly associated with neurodegenerative disease.
“This nicotine will have an impact on the PNEC, and this PNEC will release a massive amount of exosomes, and that causes perturbation in terms of iron homeostasis,” said co-first author Abhimanyu Thakur, who was with UChicago during the research and is now at Harvard Medical School’s Department of Neurosurgery. “We are finding neurodegeneration-related markers, which are going up, and which can be linked with many cognitive and dementia-related diseases.”
In simple terms, the lung cells appeared to be sending a signal that pushed neurons into mishandling iron. Too much iron in the wrong place can feed oxidative stress, strain mitochondria, and prime cells for damage. It can also contribute to ferroptosis, a form of programmed cell death driven by iron imbalance.
“This iron dyshomeostasis drives oxidative stress, mitochondrial dysfunction, and increased α-synuclein expression, hallmarks of neurodegenerative disease,” said corresponding author Asst. Prof. Joyce Chen of the UChicago Pritzker School of Molecular Engineering and the Ben May Department for Cancer Research.
The vagus nerve may help carry that message. It runs from the brain to major organs, including the lungs. It also helps regulate involuntary functions such as breathing and heart rate. Because PNECs are closely tied to lung-innervating neurons, the researchers argue that this nerve-linked network could be part of the pathway connecting nicotine exposure in the lungs to stress responses in the nervous system.
The work stretched beyond lab-grown cells. The researchers also examined mouse lungs and human lung data. They found signs that smoking or nicotine exposure was associated with more PNECs and with stronger expression of genes involved in exosome production and iron handling. In mice, nicotine exposure was also linked to poorer performance on memory and movement-related behavioral tests.
Still, the study stops short of claiming proof that smoking directly causes dementia through this pathway.
That limitation matters. The authors write that while their lab experiments support a causal role for the exosome-transferrin-TFR1 pathway in cultured cells, the evidence from human and mouse tissues remains associative. They say future experiments will need to block these exosomes, or remove the transferrin signal in living systems, to test whether this pathway truly drives disease.
They also note that several of the changes they observed reflect early vulnerability and stress, not a confirmed diagnosis of dementia itself.
Even so, the research opens a new way of thinking about the lungs.
“It reveals that the lung is not just a passive target of smoke exposure, but an active signaling organ influencing brain pathology,” Chen said.
That idea may turn out to be one of the study’s biggest contributions. For decades, smoking-related brain damage has often been framed as the downstream effect of poor circulation and chronic respiratory harm. This work adds another possibility. The lungs are not merely injured by smoke, but are actively sending damaging signals outward.
The findings could help researchers look for new ways to reduce smoking-related neurological harm before lasting damage sets in.
Blocking exosome release, interfering with the iron-transport signal, or protecting neurons from iron imbalance may eventually become targets for treatment.
The work also suggests that lung biology may deserve more attention in diseases usually treated as brain disorders alone.
Research findings are available online in the journal Science Advances.
The original story “New research finds clear link between smoking and dementia” is published in The Brighter Side of News.
Like these kind of feel good stories? Get The Brighter Side of News’ newsletter.
The post New research finds clear link between smoking and dementia appeared first on The Brighter Side of News.
Leave a comment
You must be logged in to post a comment.